Abbott Laboratories has been ordered to pay $70m in damages following a month-long trial in Cook County, Illinois, in which four families alleged the company's cow's milk-based baby formula for premature infants caused necrotising enterocolitis (NEC).
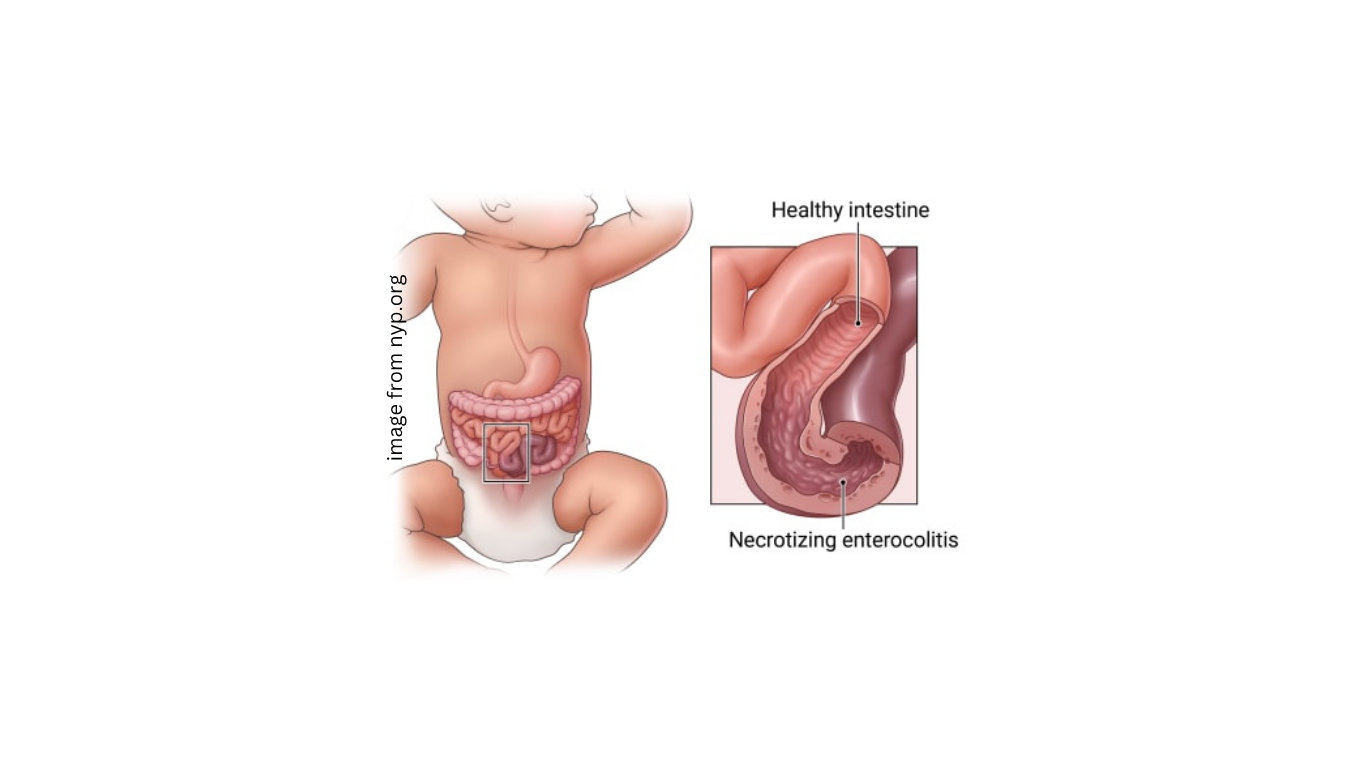
NEC is a severe and potentially fatal bowel disease primarily affecting preterm newborns.
The jury found Abbott liable on three counts: product defect, failure to warn and negligence. It awarded $53m in compensatory damages to the four children, all born before 32 weeks' gestation at Chicago-area hospitals between 2012 and 2019, who developed NEC after consuming Abbott's Similac Special Care.
A further $17m in punitive damages was added on Friday after jurors determined Abbott's conduct was "willful and wanton." Three of the four children required surgery; all four continue to experience gastrointestinal complications.
Abbott corporation maintains that Similac Special Care is an essential product for neonatal intensive care units when maternal or donor breast milk is unavailable.
The scientific picture remains contested. A 2024 joint report from US regulatory agencies and a National Institutes of Health working group concluded that current evidence supports the hypothesis that the absence of breast milk, rather than exposure to cow's milk-based formula, is associated with increased NEC incidence.
Abbott's CEO has previously indicated the company may withdraw its preterm formula products altogether if litigation pressure continues, which is a prospect that neonatal clinicians have warned could leave hospitals with critically limited feeding options.
The verdict is the second trial loss for Abbott in this litigation.
A St Louis jury previously ordered the company to pay $US495 million in a separate NEC case, which is currently under appeal.
Abbott and co-defendant Mead Johnson (the manufacturer of Enfamil preterm formula) now face nearly 1,000 combined lawsuits, with more than 700 consolidated in Illinois federal court.
No federal cases have yet proceeded to trial.
The outcome is likely to intensify scrutiny of product labelling standards, risk communication obligations and requirements for formulation safety evidence.
This article was adapted from https://manufacturingchemist.com/abbott-70-million-damages-verdict-preterm-infant-formula






Laisser un commentaire
Ce site est protégé par hCaptcha, et la Politique de confidentialité et les Conditions de service de hCaptcha s’appliquent.